Covalent Diamond–Graphite Bonding: Mechanism of Catalytic Transformation

Semir Tulić, Thomas Waitz, Mária Čaplovičová, Gerlinde Habler, Marián Varga, Mário Kotlár, Viliam Vretenár, Oleksandr Romanyuk, Alexander Kromka, Bohuslav Rezek, Viera Skákalová
ACS Nano, 2019, 1344621-4630
https://doi.org/10.1021/acsnano.9b00692
Abstract
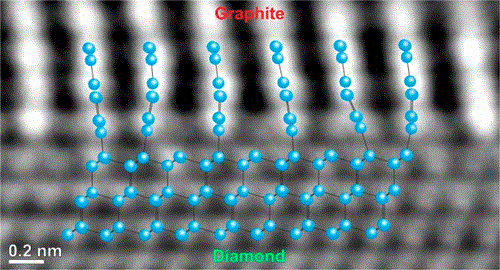
Aberration-corrected transmission electron microscopy of the atomic structure of diamond–graphite interface after Ni-induced catalytic transformation reveals graphitic planes bound covalently to the diamond in the upright orientation. The covalent attachment, together with a significant volume expansion of graphite transformed from diamond, gives rise to uniaxial stress that is released through plastic deformation. We propose a comprehensive model explaining the Ni-mediated transformation of diamond to graphite and covalent bonding at the interface as well as the mechanism of relaxation of uniaxial stress. We also explain the mechanism of electrical transport through the graphitized surface of diamond. The result may thus provide a foundation for the catalytically driven formation of graphene–diamond nanodevices.
